Hydroquinone: Everything You Need To Know
Hydroquinone is a highly controversial ingredient found in skin lightening creams around the world.
In 2006, the United States Food and Drug Administration (FDA) proposed to eliminate over-the-counter (OTC) skin bleaching creams with up to 2% hydroquinone. (1)
The FDA cited safety concerns including the potential risk of cancer and multiple reports of exogenous ochronosis (i.e. skin darkening) associated with hydroquinone.
Internationally, hydroquinone for OTC use has been banned in The European Union, Australia, and Japan, and prohibited (or restricted) in several African countries including Rwanda and Ghana. (2)
So it's no wonder that many consumers are now asking, "is hydroquinone safe?"
In this article, I've prepared a full summary of the medical data on hydroquinone to help you weigh the risks and rewards appropriately so that you can make an informed decision.
RELATED ARTICLE:
RELATED ARTICLES:
- 7 Best Body Sprays for Acne Prone Skin of 2026 (Ranked & Reviewed)
- 7 Best Serums for Dark Spots and Hyperpigmentation of 2026 (Ranked & Reviewed)
What Is Hydroquinone?
Hydroquinone is a white, crystalline solid with the chemical formula C6H4(OH)2.
Discovered in 1843, it's primarily used today in skin lightening creams and photographic developers.
Hydroquinone is produced through chemical processes starting with either benzene or phenol.
How Does Hydroquinone Work?
In your skin, there are special cells called melanocytes located in the stratum basale (the basal layer or "basement" of the epidermis).
Melanocytes produce an important pigment, known as melanin, that gives your skin its natural color and protects it from free radical damage, such as ultraviolet radiation from the Sun.
Melanin pigments are further packaged into melanosomes (a bulk storage container for melanin) and then distributed to keratinocytes ("rank-and-file" skin cells that make up 90% of the epidermis).
Within each keratinocyte, the melanosomes form a protective "shield" around the nucleus (where your DNA resides) to prevent UV-induced DNA mutations that may lead to skin cancer.
In other words, melanin is very important for skin health.
However, too much melanin will lead to unwanted dark spots and uneven skin tone.
This can be triggered by a multitiude of factors, including hormonal changes (such as pregnancy), prolonged sun exposure, and inflammatory skin diseases such as acne (i.e. post-acne marks).
Enter Hydroquinone.
Hydroquinone improves hyperpigmentation by slowing down the process of melanin production. It does this by reducing the effectiveness of an enzyme called tyrosinase (required to synthesize melanin).
In fact, hydroquinone is considered one of the most effective tyrosinase inhibitors and often called the "Gold Standard" for hyperpigmentation issues such as melasma.
The Hydroquinone Controversy
Since the 1950s, hydroquinone has been used in skin bleaching creams around the world.
In 1982, the FDA approved the use of up to 2% hydroquinone in OTC skin bleaching creams and tentatively labeled it as "Generally Recognized As Safe and Effective (GRASE)". (1)
However, new studies in the following decades prompted the FDA to do a full 180° turn. In 2006, the agency proposed to ban OTC skin bleaching creams with hydroquinone altogether.
So why the (not so sudden) change of heart?
The FDA's main concerns with topical hydroquinone usage is two-fold:
- There is "some evidence" that hydroquinone can cause cancer
- There have been reports of exogenous ochronosis following prolonged use of hydroquinone (mostly in South Africa)
In response to the proposed rule, several organizations have opposed the FDA's decision, labeling the move as "extreme" given the clinical evidence to date. (1)
These include highly respected professional associations such as the American Academy of Dermatology Association and American Health & Beauty Aids Institute.
So who should you trust in this matter?
As I always say, "show me the evidence!"
The Medical Data on Hydroquinone
The FDA outlined four main areas of concern regarding hydroquinone safety:
- Fertility
- Absorption
- Cancer
- Exogenous Ochronosis
Hydroquinone & Fertility
"The FDA noted that existing studies involving the effects of hydroquinone on fertility have produced conflicting results." (1)

Studies show that hydroquinone may have an impact on fertility and pregnancy:
- In 1985, the Environmental Protection Agency (EPA) cited two studies where hydroquinone prolonged the estrous cycle of female rats and reduced the fertility of male rats. (3)
- Salzgeber (1955) found that hydroquinone inhibited the normal growth of ovaries in 7 out of 15 chick embryos grown in vitro. However, Burgaz et al. (1994) noted limited effects from injecting hydroquinone into chick embryo air sacs compared to controls. (1)
- Telford et al. (1962) reported that adding hydroquinone to the diet increased the incidence of unsuccessful pregnancies in female rats to 100% vs. 40.8% of controls. But a similar study submitted by the Chemical Manufacturers Association showed no adverse effects. (1)
- A study of pregnant rabbits found no impact at low doses of hydroquinone, limited impact at medium doses, and increased incidences of fetus malformations at high doses. (1)
- In a literature review by DeCaprio (1999), the author noted that dermal application of hydroquinone had no negative effects on pregnant rats. (4)
- The FDA also cited a study where hydroquinone was administered to two consecutive generations of rats. No adverse effects were reported in either generation. (1)
In summary, the FDA concluded that "[it] cannot make a final determination on hydroquinone's potential to impair fertility [until further studies are conducted]."
So what does it all mean, in English?
In my opinion, these studies indicate that:
Oral consumption of hydroquinone at high concentrations harms both fertility and overall health.
But...
Skin lightening creams are, of course, applied on your skin (not indigested). In addition, OTC products in the US are restricted to using low concentrations of hydroquinone (2% or less).
So the real question is, "is hydroquinone safe when applied topically at low concentrations?"
To find out, we'll need to examine the absorption rate of hydroquinone in human skin.
Hydroquinone & Absorption
"The rate of absorption [...] by human skin caused the [FDA] to recommend additional studies using OTC concentrations of hydroquinone." (1)

The FDA cited two studies on hydroquinone absorption in human skin:
- Bucks et al. (1988) found that 2% hydroquinone in an alcoholic base readily penetrated human forehead skin with an average absorption rate of 57% after 24 hours. Applying sunscreen significantly reduced absorption to only 26%. (5)
- Wester et al. (1998) applied a 2% hydroquinone cream on the forearms of volunteers and noticed rapid skin penetration. Hydroquinone was detected in blood samples within 30 minutes with peak levels occurring after 4 hours. (6)
So again, in English?
Hydroquinone is quickly and thoroughly absorbed by your skin.
Based on these studies, it can be absorbed in as little as 30 minutes with almost 60% absorption after 24 hours.
This raises the concern of adverse side effects since hydroquinone can be toxic at high concentrations and is a possible carcinogen (the evidence is still unclear). As the FDA stated:
"The evidence of [cancer] in animals in combination with the high absorption rate (57%) of hydroquinone [...] does not allow FDA to rule out [the risk of cancer in humans]." (1)
The high absorption rate might explain also why hydroquinone is so effective at treating hyperpigmentation, such as melasma, and for general skin lightening purposes.
It can penetrate your skin to reach the deepest levels of the epidermis (stratum basale) where it inhibits melanocyte activity (the cells that produce melanin and cause dark spots).
Hydroquinone & Cancer
"Based on the evidence of [cancer] in animals, FDA cannot rule out the potential [cancer] risk from topically applied hydroquinone in humans. (1)"

Now, for the question we've all been waiting for: "can hydroquinone cause cancer?"
Unfortunately, there's no definitive answer.
But wait! Before you click the "back" button, allow me to explain:
- The FDA says there is "some evidence" that hydroquinone causes cancer based on the results of a 2-year study by the National Toxicology Program in 1989
- The results from this study did not meet the FDA's requirements for "clear evidence" (i.e. the connection between hydroquinone and cancer is still unclear)
- Moreover, no hydroquinone-related cases of cancer have been reported in humans
As you can see, it's extremely difficult to provide a "yes" or "no" answer to this question.
Some sites, like Healthline, deem hydroquinone safe but fail to discuss the FDA's safety concerns or the fact that hydroquinone is already banned in the European Union, Japan, and Australia.
So rather than making a definitive statement, I think it's better to show you the data and let you weigh the risks and rewards of using hydroquinone according to your situation.
Of course, if you have any concerns, you should seek medical advice from a licensed physician.
Now, let's take a deeper look at this 2-year study by the National Toxicology Program (NTP).
In this study, researchers fed hydroquinone to four groups of rodents (male & female / rats & mice) for 5 days a week over a period of 2 years.
Each group was split into a control group (no hydroquinone), low dose group (25 mg/kg), and high dose group (50 mg/kg). (7)
By the end of the study, the NTP concluded there was "some evidence" of cancer:
- Male Rats: test groups experienced higher incidences of benign kidney tumors (7% in low dose and 14% in high dose compared to 0% in controls). Hyperplasia (abnormal cell growth that may lead to cancer) was observed in 2 out of 55 rats in the high dose group.
- Female Rats: test groups experienced significantly greater cases of leukemia (27% in low dose and 40% in high dose compared to 16% in controls).
- Male Mice: test groups showed higher incidences of benign liver tumors (39% in low dose and 36% in high dose compared to 16% in controls) but this was offset by fewer cases of cancerous liver tumors (20% in low dose and 13% in high dose compared to 24% in controls).
- Female Mice: test groups exhibited higher incidences of mainly benign liver tumors (29% in low dose and 24% in high dose compared to 5% in controls).
The FDA's Carcinogenicity Assessment Committee reviewed the NTP study and agreed that there is "some evidence" of cancer in male and female rats and female mice.
However, some researchers have remained skeptical about the NTP study's findings:
- Hard et al. (1997) and McGregor (2007) reexamined the kidney sections of male rats and concluded that hydroquinone did not directly cause tumors, but rather exacerbated an existing kidney disease that affects only rats with no known counterpart in humans. (2)
In conclusion, it's still unclear whether prolonged use of skin bleaching creams with hydroquinone can cause cancer.
Based on "some evidence" from animal studies, the FDA has not ruled out the possibility of cancer in humans and has recommended additional studies on the topic.
Hydroquinone & Exogenous Ochronosis
"Further, hydroquinone has been shown to cause disfiguring effects (ochronosis) after use of high concentrations (5% or greater) and at concentrations as low as 1 to 2%." (1)
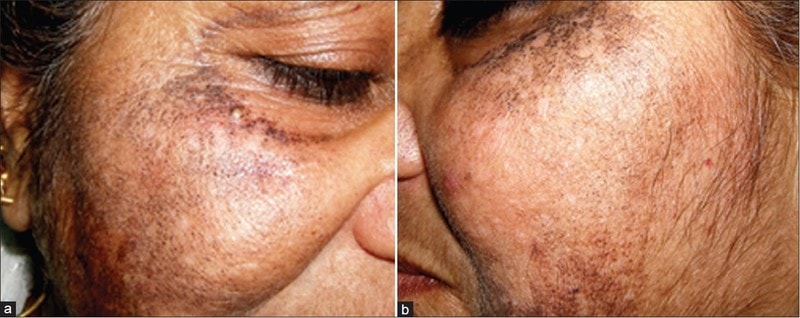
Prolonged use of skin bleaching creams with hydroquinone has been associated with a skin condition known as exogenous ochronosis, particularly in South Africa.
This condition is characterized by deposits of blue-black pigments on the skin that result in skin darkening, rather than the desired skin lightening effects of hydroquinone.
Findlay et al. (1975) first documented exogenous ochronosis in South African women who had been using a high concentration of hydroquinone (5% or higher) for over 3 years. (1)
The FDA reviewed this study and concluded that:
"[...] prolonged use of high concentrations (5% or more) of hydroquinone [along] with exposure to sun may produce disfiguring effects.' (1)
According to a literature review, 789 cases of exogenous ochronosis have been reported worldwide, of which 756 were in South Africa and 22 were in the US. (8)
Researchers have presented several reasons for the greater prevalence in South Africa (1):
- Higher concentrations of hydroquinone were used before 1984
- Additives such as t-butyl alochol and mercury were common before 1986
- Hydroalcoholic lotion is often added to increase skin penetration
- Resorcinol in often used in combination with hydroquinone
- The climate is mostly sunny and people tend not to use sunscreen as regularly
While severe cases of exogenous ochronosis can be disfiguring, Levitt (2007) notes that the incidence rate is very low, with only 22 reported cases in the US in more than 50 years. (8)
Overall, exogenous ochronosis is a potential side effect of using hydroquinone but may be avoided by taking these safety precautions:
- Apply a broad-spectrum sunscreen with at least SPF 50 when heading outdoors
- Avoid using skincare products that increase sun sensitivity or skin penetration at the same time
- Be aware that prolonged use of hydroquinone, rather than a high concentration, is a key contributing factor to exogenous ochronosis
Current Regulations on Hydroquinone
Hydroquinone has been banned from OTC use (or heavily restricted) in the following regions:
The following countries have proposed to limit over-the-counter hydroquinone products:
In South Africa, only 2% hydroquinone products are permitted and must include at least SPF 5.
In Canada, hydroquinone above 2% is available by prescription only.
Conclusion: Should You Use Hydroquinone?
Ultimately, the decision is up to you as skin bleaching creams with hydroquinone remain widely available around the world.
Hopefully, this article has shed more light on the potential risks of hydroquinone to help you make the best decision for your skin health.
In many respects, the clinical evidence is simply unclear and experts from regulators to dermatologists to industry associations disagree on the best policy for hydroquinone usage.
What has been your experience with hydroquinone? Let me know in the comments!
Save Article on Pinterest

Sources
(1) Proposed Rule on Skin Bleaching Drug Products For Over-the-Counter Human Use. The United States Food and Drug Administration. Federal Register (2006).
(2) Supporting Information for Toxicological Evaluation by the National Toxicology Program. Prepared by the Food and Drug Administration. National Toxicology Program (2009).
(3) Testing Requirements for Hydroquinone. United States Environmental Protection Agency. Federal Register (1985).
(4) The Toxicology of Hydroquinone. DeCaprio AP. Critical Reviews in Toxicology (1999).
(5) Percutaneous Absorption of Hydroquinone in Humans. Bucks DA, et al. Journal of Toxicology and Environmental Health (1988).
(6) Human in vivo and in vitro Hydroquinone Topical Bioavailability, Metabolism, and Disposition. Wester RC, et al. Journal of Toxicology and Environmental Health (1998).
(7) NTP Toxicology and Carcinogenesis Studies of Hydroquinone. National Toxicology Program. National Toxicology Program Technical Report Series (1989).
(8) The Safety of Hydroquinone: A Dermatologist's Response to the 2006 Federal Register. Levitt J. Journal of the American Academy of Dermatology (2007).


